We are facing an antibiotic resistance crisis. Almost every health authority has sounded the alarm and the most recognized authority, the World Health Organization, is doing all it can to slow the arrival of the post-antibiotic era. Yet, even as these calls are made, the use of these drugs both in medicine and agriculture continues to be unacceptably high. For public health officials, this represents trouble as more bacteria gain the ability to withstand several, if not all, treatments.
Several approaches to tackling these infections are needed. One route is to develop stronger antibiotics, which is ongoing. But there are other less pharmaceutical-based options. These include the use of bacterial viruses, known as bacteriophages, and molecules known to kill bacteria called antimicrobial peptides.
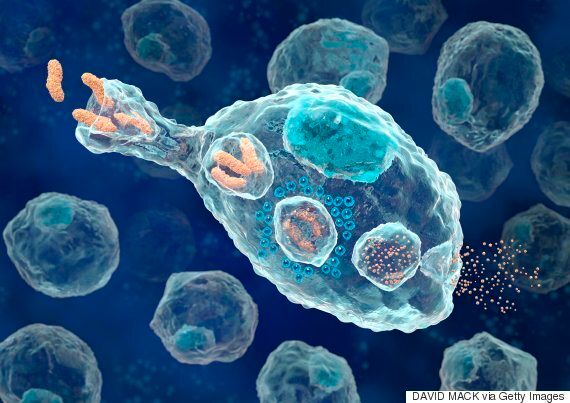
A macrophage, seen here engulfing bacteria, is a white blood cell which attacks foreign organisms (pathogens) in the body. The pathogens are broken down and destroyed once they have been engulfed, and the remaining parts are ejected (far right) by the cell. (Photo: David Mack via Getty Images)
Now there may be a new possibility, thanks to a group of European researchers. They have developed a unique approach in which treatment is not based on drug administration, but rather the use of molecules found inside our own bodies. Their findings suggest the use of special types of fat molecules may provide a unique means to deal with resistance.
The team focused on a particular cellular member of our immune system. It's called a macrophage. One of its primary goals is to eat foreign particles, a process known as phagocytosis, and break them up inside the cell. When a bacterial infection begins, these cells are sent to the area to swallow as many as possible to reduce the chances of disease.
Macrophages also have another important role. They act as waste collectors in the body. They look for signs of recently deceased cells, known as apoptotic bodies. As with any waste, accumulation can become toxic to the body. The macrophages eat up and digest the cellular remnants to keep us safe.
There is an added advantage to this waste removal process. After macrophages ingest these apoptotic bodies, they become better defenders against bacteria. It's because the presence of dead cellular fragments suggest there may be a microbial invasion. Though this may not be the case for the immune system, being prepared is always best.
Research into the mechanism behind this defence enhancement has revealed the central molecules are not proteins as one might expect. Instead, it occurs due to the presence of lipids, which we generally called fats. As the macrophage breaks down apoptotic bodies, it identifies certain types of fats as important and protects them from digestion. These redeemed molecules are sent to various areas of the cell known to be responsible for killing bacteria. When they arrive, the cellular soldier becomes a better bacterial killer.
For the European researchers, this additional benefit was worth investigating further. If they were correct, the delivery of these fats to macrophages would lead to an improved defence against bacteria, including antibiotic-resistant species. But to get to this result, they needed to test their theory in the lab.
If they were correct, the delivery of these fats would lead to an improved defence against bacteria, including antibiotic-resistant species.
Using laboratory cultures of macrophages, the team introduced specially made apoptotic bodies containing the special types of lipids. As expected, the macrophages changed and became better prepared to fight. When bacteria were introduced into the system, they were attacked within hours. The results confirmed the group's hopes, at least in the laboratory model.
The next stage of the experiment was to examine the effects of the specialized fats on cells taken from people suffering from pneumonia. A total of six patient samples were used -- this was a preliminary trial after all. The samples were taken back to the lab for analysis and testing.
The first step was to identify the species causing the trouble and more importantly, if any were resistant to antibiotics. A total of seven different pathogens were collected from the samples. Several were antibiotic resistant and in one case, the species was able to resist all but one available antibiotic. For the team, this was the prefect challenge.

The samples were given the fats in the form of those apoptotic bodies. Then the team had to wait 18 hours. If they were right, the levels of bacteria would be reduced. If they were wrong, they would have to admit defeat.
Thankfully, the results were better than expected. Compared to controls, the bacterial levels were reduced dramatically. In some cases, the addition of the fats led to a 99 per cent decrease in the bacterial population. Even more important was the fact the most affected species happened to be the one resistant to so many antibiotics. The trial was a complete success.
Although the team studied human samples, don't expect to hear of this fat-based treatment anytime soon. The use of fats to combat antibiotic resistant bacteria need to undergo many more stages of testing before they can be used in clinical trials. Yet, these experiments offer some great promise to deal with the crisis in the future.
In the meantime, we still can help slow down the pace of antibiotic resistance. First and foremost, we can reduce the need for these drugs when we feel sick. Instead of asking for them when we see the doctor, we can allow for the proper testing to be performed and only receive a prescription when necessary. We also can seek out meats from animals raised without antibiotics. Their presence in grocery stores has been growing significantly and the price difference for the most part has lessened.
Follow HuffPost Canada Blogs on Facebook
Also on HuffPost:
